
 About Us
About Us
Tooba Pharmaceuticals is a WHO-GMP Certified Manufacturer of API Bulk Drugs and Intermediates with a focus on quality, innovation, and compliances. With a state-of-the-art manufacturing facility and a commitment to excellence, we cater to the global pharmaceutical industry.
World-class infrastructure and quality systems adhering to global standards.
Strict adherence to regulatory norms ensures customer confidence worldwide.

 Products
Products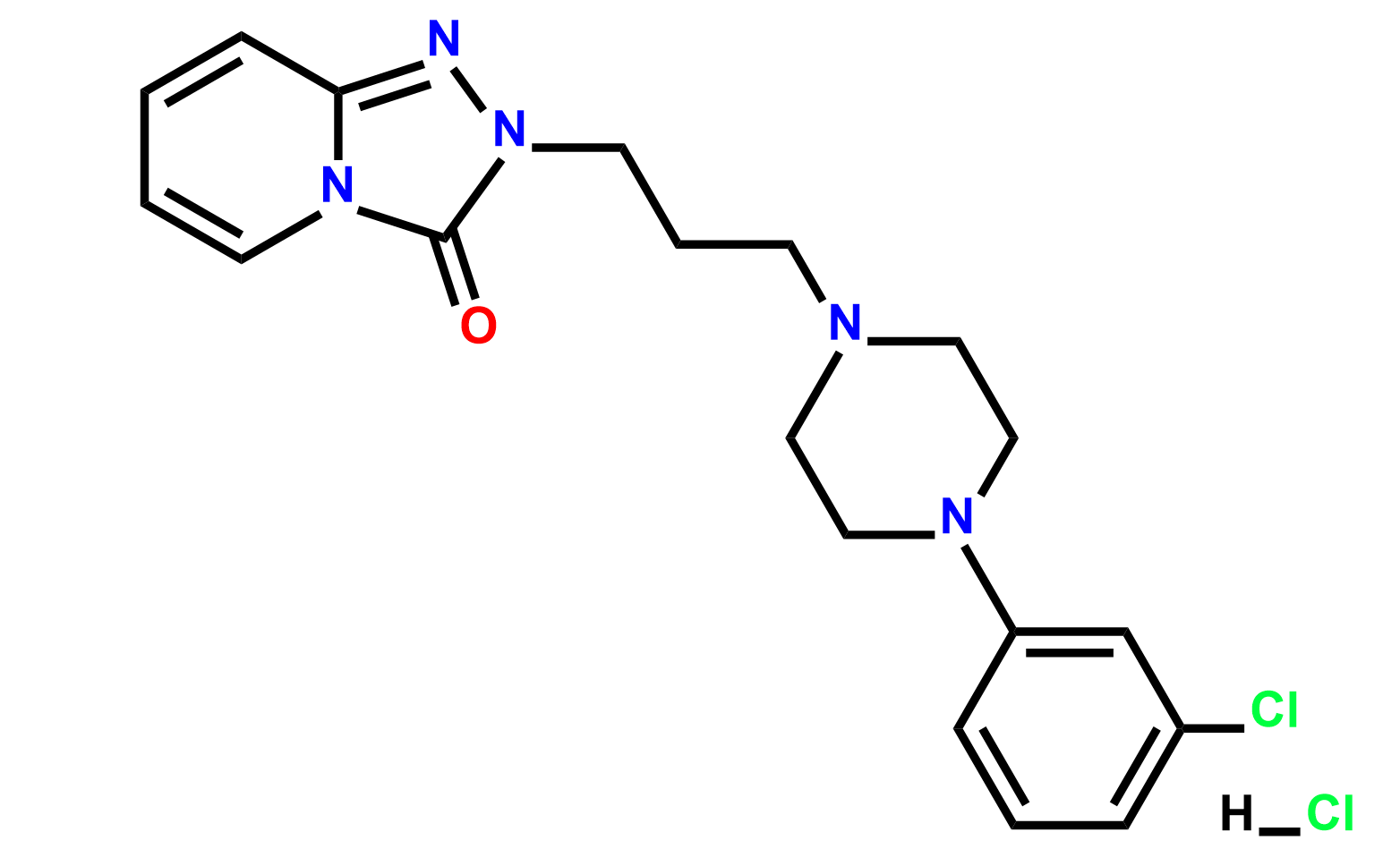
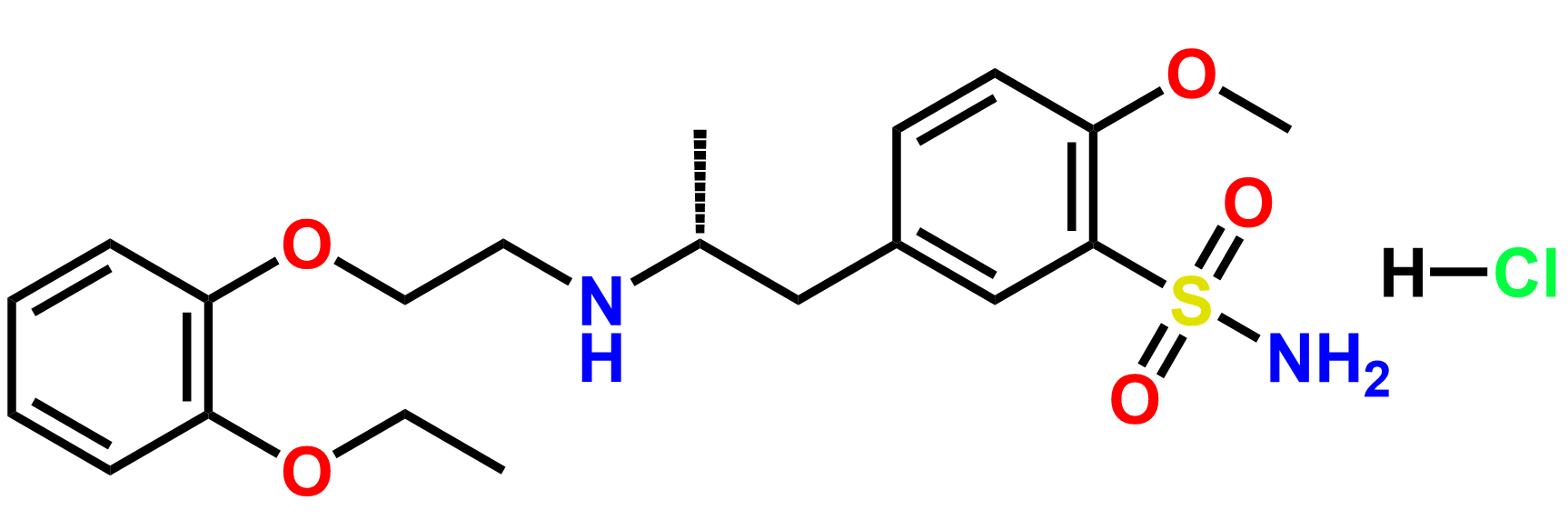
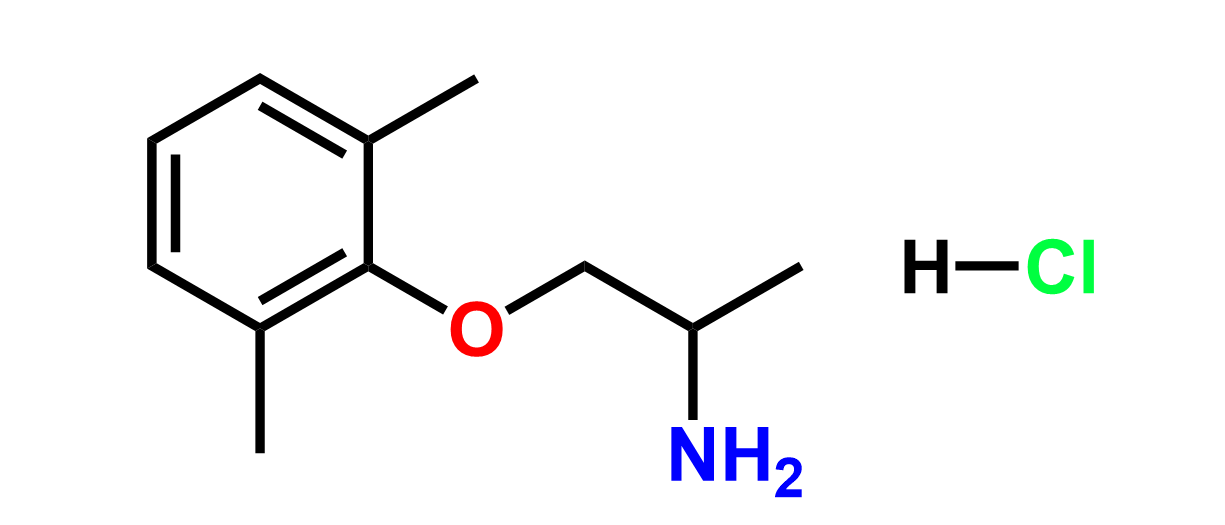
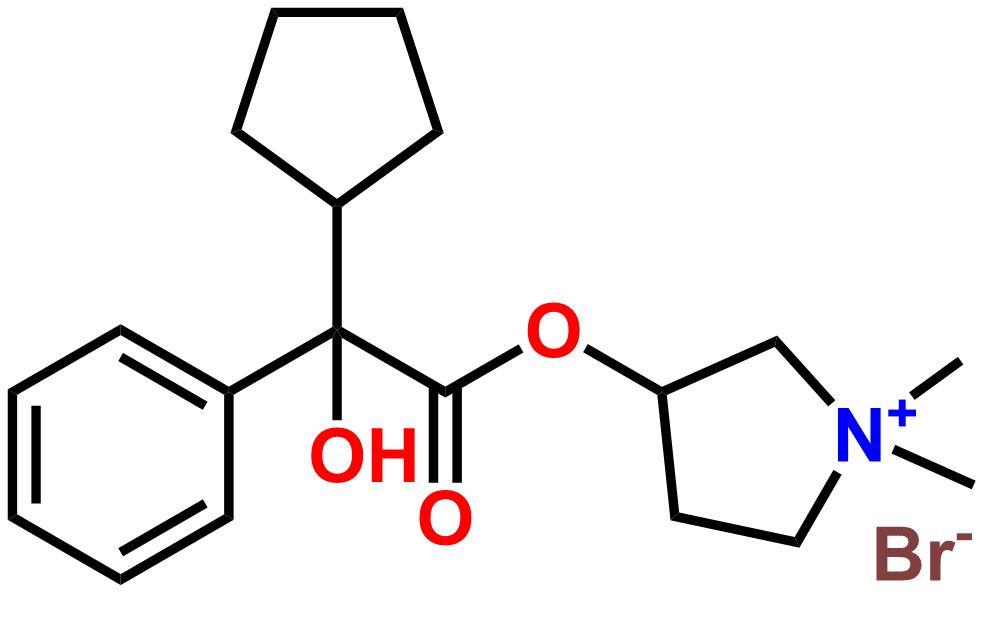
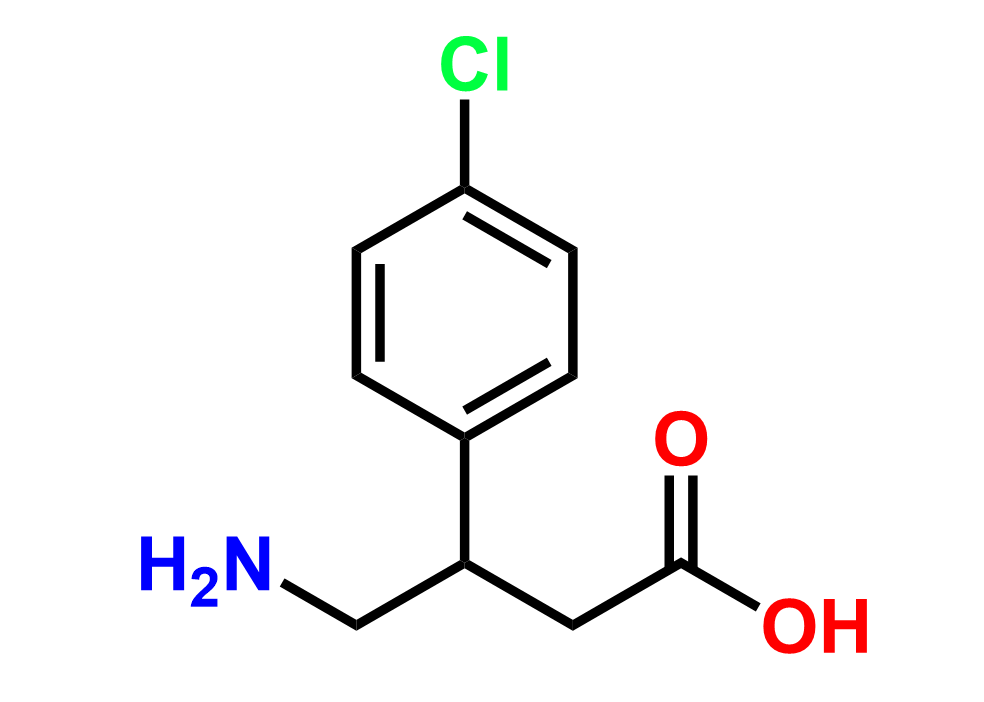
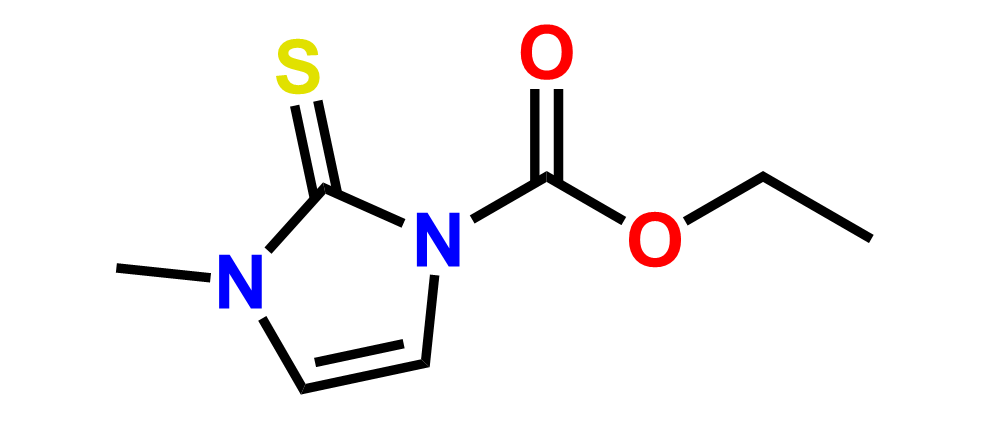
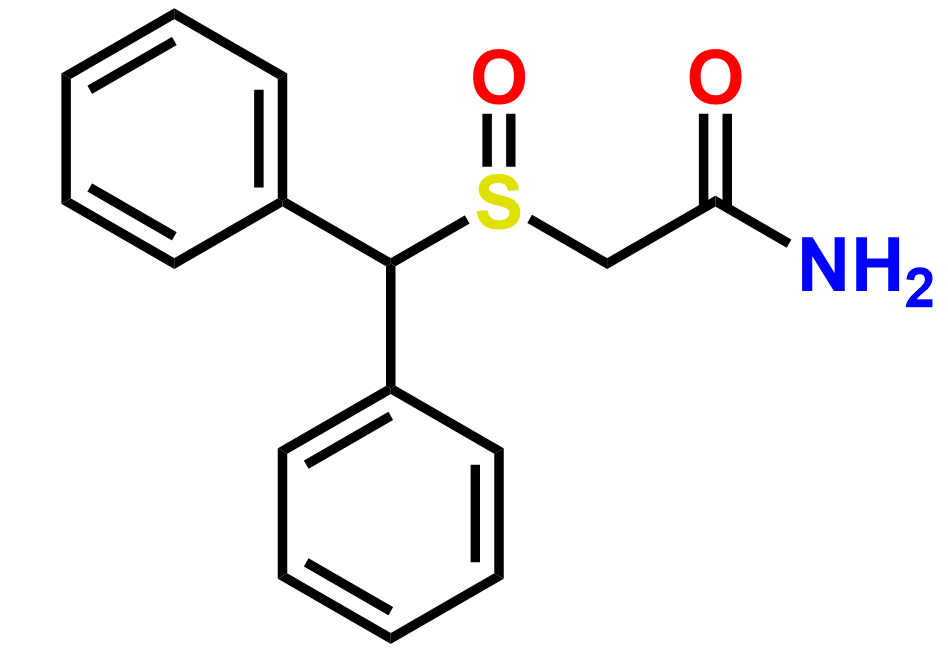
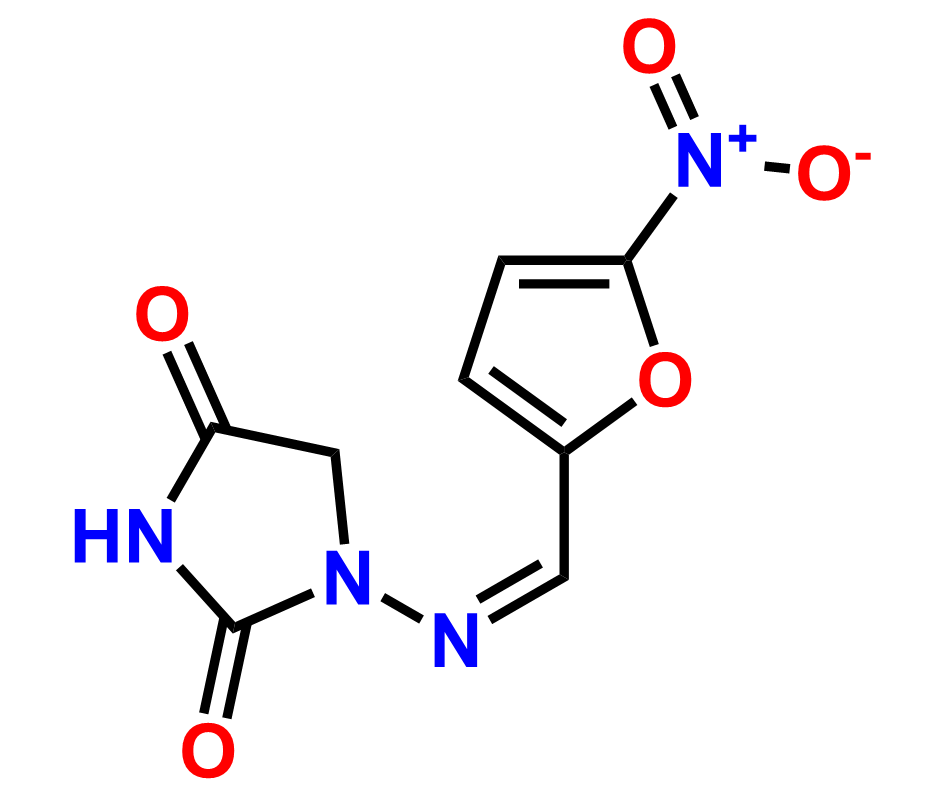
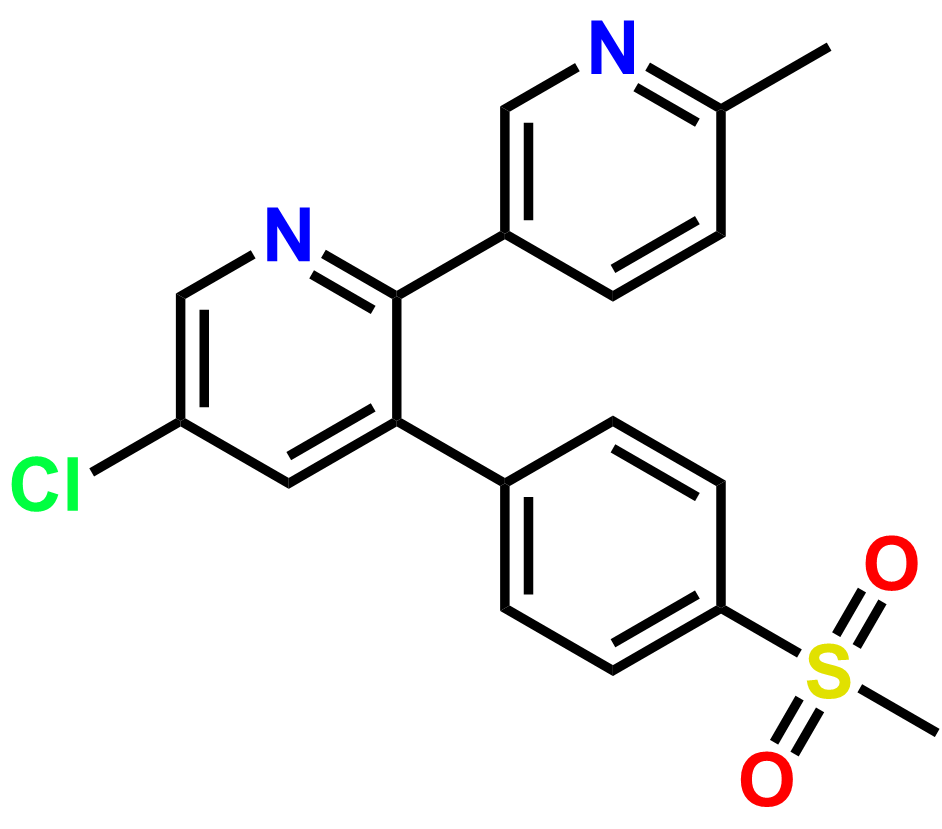
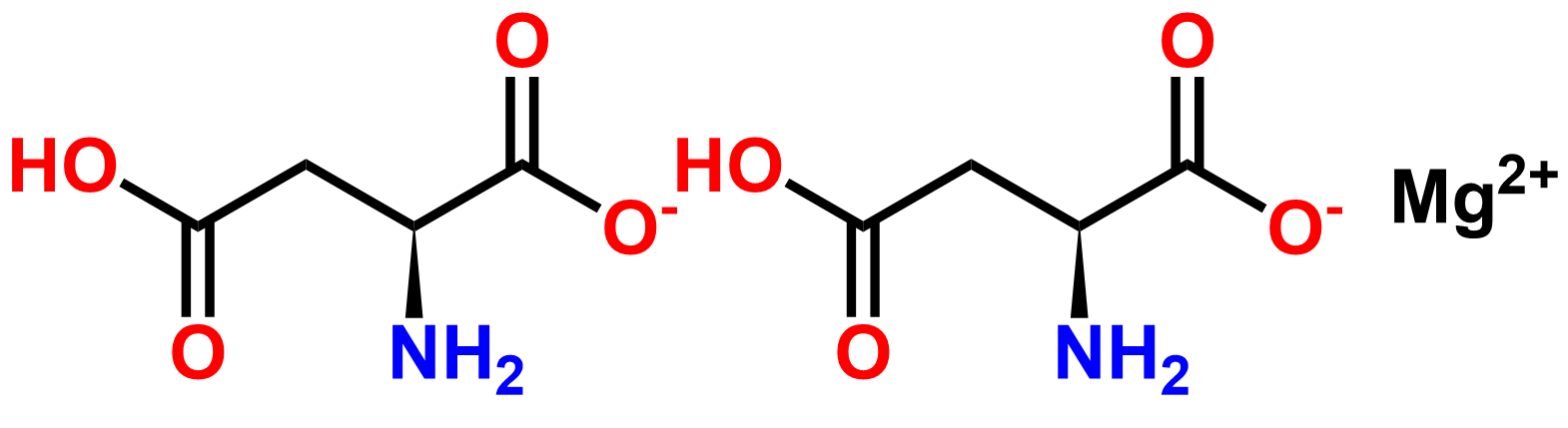
 Why choose us
Why choose usAt Tooba Pharmaceuticals, we are committed to delivering high-quality APIs backed by innovation, regulatory compliance, and customer-centric services. Here's why we stand out:

We are a WHO-GMP certified manufacturer of Active Pharmaceutical Ingredients (APIs), ensuring consistent quality, reliability, and regulatory compliance.
We have filed a patent (Application No. 202021022784) for the process of Glycopyrrolate and its purification—demonstrating our commitment to continuous innovation and superior product quality and patents filing for other APIs are also planned.

We offer complimentary available samples of both Impurity Working Standards and API Working Standards with order or as and when required to support your analytical and testing needs.

Our fully equipped QMS provides all necessary documents for vendor qualification and regulatory approvals.

All our APIs are tested and compliant with the latest editions of relevant pharmacopoeias (USP, Ph. Eur., IP, etc.).

We have conducted a detailed N-Nitrosodimethylamine (NDMA) risk assessment to ensure the highest safety standards for our products.

We support on-site customer audits at our expense or arrange virtual audits at your convenience after commercial confirmations.
Years
Cumulative R&D Experience
Patents Credited
to Our
Experts
QMS
Documentation Support
Instrument &
Equipment
 Tooba Pharma
Tooba Pharma



